A Cleveland Clinic research team is developing computational representations of the human body in order to understand how movement patterns and loads on the joints deform the surrounding tissues and cells. Utilising the computing systems of the Ohio Supercomputer Center in the US, the team hopes the virtual models will someday be used to understand damage mechanisms caused by the aging process or by debilitating diseases, such as osteoarthritis.
Leading the team is Ahmet Erdemir, director of the Computational Biomodeling Core (CoBi) and a faculty member in the Department of Biomedical Engineering at the Lerner Research Institute (LRI) in Cleveland, Ohio. ‘The aging process and debilitating diseases affect many aspects of the mechanical function of the human body, from the way we move to how our muscles, joints, tissues, and cells accommodate the loading exerted on the body during daily activities,’ Erdemir explained. ‘Computational modelling techniques provide an avenue to obtain additional insights about mechanics at various spatial scales.’
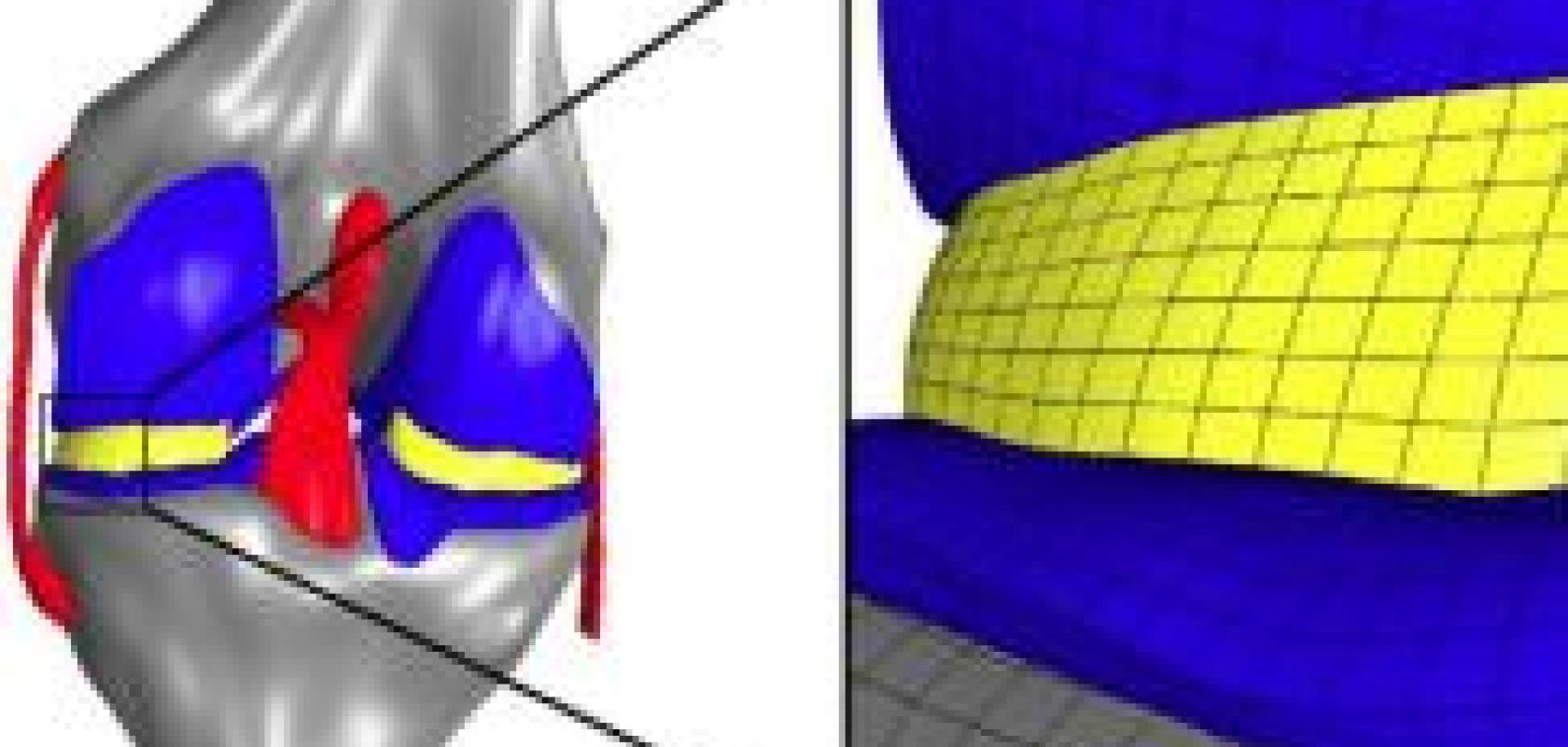
While many macro-scale studies have looked at how the various components of a knee joint respond to weight and other external loads, Erdemir and colleague Scott C. Sibole wanted to better understand how those large mechanical forces correspond to the related deformation of individual cartilage cells – or chondrocytes – within the knee. Previous micro-scale studies of cartilage have not commonly been based on data from body-level scales, in particular, by the musculoskeletal mechanics of the knee joint. Furthermore, calculated deformations have typically been for a single cell at the centre of a 100-cubic-micrometre block of simulated tissue; Erdemir used an anatomically-based representation that calculated deformations for 11 cells distributed within the same volume.
Erdemir’s method proved to be highly scalable because of micro-scale model independence that allowed exploitation of distributed memory computing architecture. As a result, Sibole, a research engineer at LRI, was able to harness the 75 teraflops of computational power of OSC’s IBM 1350 Glenn Cluster. When engineers recently deployed the centre’s more powerful HP-Intel Xeon Oakley Cluster, the Glenn Cluster was partially decommissioned.
An article authored by Erdemir and Sibole, ‘Chondrocyte Deformations as a Function of Tibiofemoral Joint Loading Predicted by a Generalized High-Throughput Pipeline of Multi-Scale Simulations’, was recently published in PLoS ONE, an international, peer-reviewed, open-access, online journal. The article can be viewed at: http://dx.doi.org/10.1371/journal.pone.0037538. Grant funding from the National Institute of Biomedical Imaging and Bioengineering, National Institutes of Health supported the study.