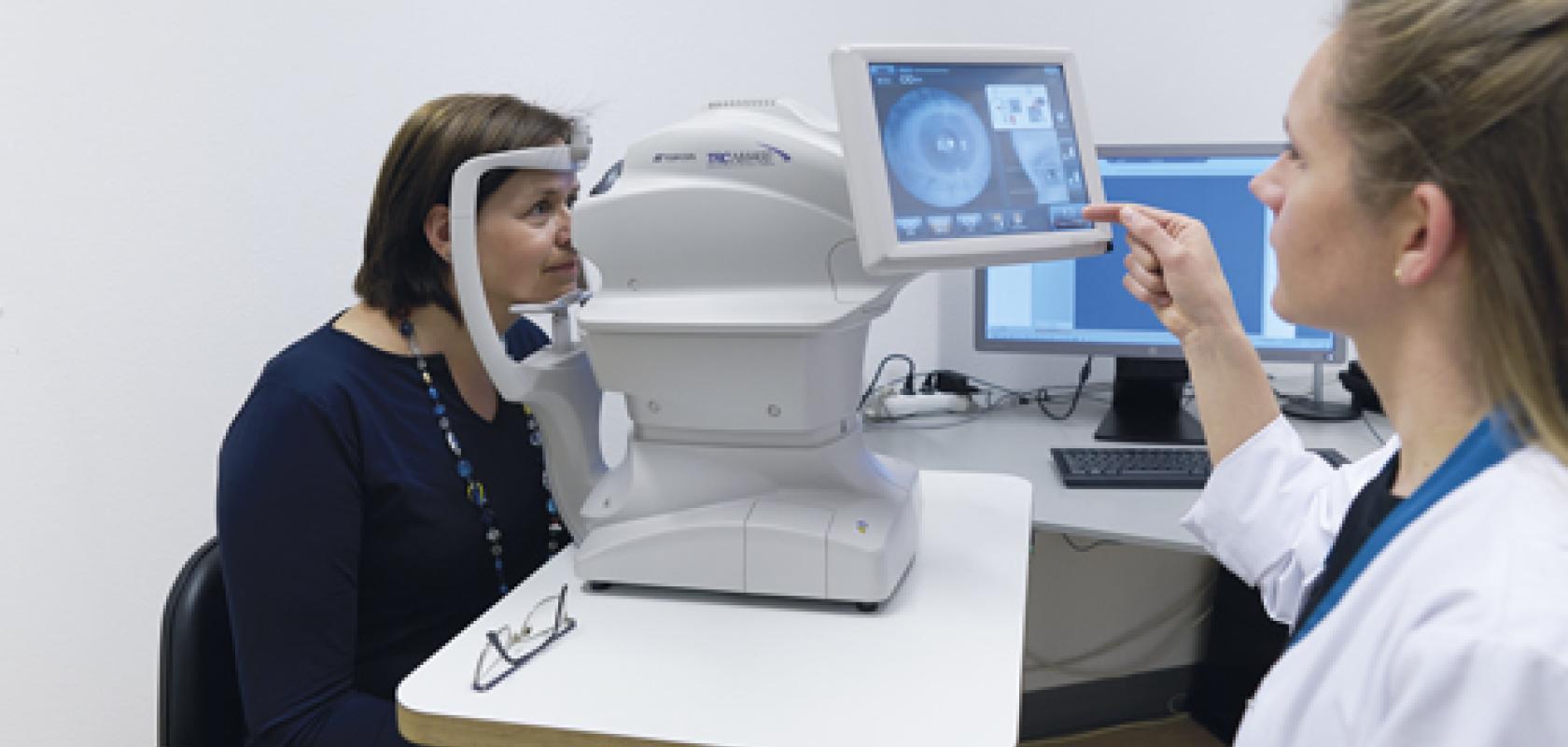
In January 2020 the American Diabetes Association released a new set of clinical standards that, for the first time, included the use of an FDA-approved autonomous artificial intelligence (AI) platform for detecting diabetic retinopathy in people with diabetes.
The system, IDx-DR, has been developed by Iowa-based IDx Technologies, and can carry out a clinical diagnosis from retinal images, in real time. FDA approved the autonomous AI technology that underpins IDx-DR in April 2018, through a 510k de novo authorisation process.
And it’s the term autonomous that’s key here. ‘Our system is the only autonomous AI, in any industry, that is available to the public in the US,’ commented Michael Abramoff, founder and executive chairman of IDx. ‘You can’t yet buy a self-driving car and take it on public roads, and you won’t get a loan decision from an AI software without a human overseeing it. As a completely autonomous AI system, IDx-DR is FDA-approved to make clinical decisions without any human oversight whatsoever.’
The analysis for diabetic retinopathy is carried out on two retinal images per eye, obtained using a standard fundus camera. The images are transferred by the operator, via computer, to the IDx-DR analysis system. However, rather than being assessed by an ophthalmologist or specially trained practitioner, the images are analysed solely by the IDx-DR platform – which, within seconds, provides either a positive or negative clinical decision, together with follow-up care instructions.
If the images are of sufficient quality, the software provides one of two results: either ‘more than mild diabetic retinopathy detected: refer to an eye care professional’, or, ‘negative for more than mild diabetic retinopathy; rescreen in 12 months.’ If the image quality isn’t high enough, the system guides the operator through retaking an image.
The system can directly interface with other medical software and healthcare records, so it can be implemented rapidly into healthcare environments where patients may otherwise have to wait months before an appointment with an ophthalmologist becomes available, comments Abramoff.
It’s because IDx-DR is the first completely autonomous AI to be cleared for public use that the regulatory approval process has thrown up a whole raft of clinical trials-related, regulatory, and insurance-related considerations and hurdles – technological factors aside – that have taken Abramoff and his company nearly 10 years to negotiate.
‘We had to work closely with FDA to secure the most appropriate testing route for approval, as well as work with the US healthcare system and insurance providers,’ Abramoff noted. Issues around insurance have been particularly interesting. ‘We assume full liability for the AI’s performance – just as a physician has for their decisions - so it was of huge interest to all developers of autonomous AI technologies, and not just those in the healthcare field, to see how we would be insured, what is the chain of liability, etc. and how potential litigation may play out.’
Getting a truly autonomous AI system into the clinical decision-making arena was a huge achievement, Abramoff acknowledged, not just because of the insurance issue. FDA had never approved an autonomous AI system, so this was breaking new ground on many fronts. ‘We had two options, then, for our first market approval,’ Abramoff noted. ‘We could go to a country with a less stringent regulatory process, perhaps in the developing world, get the experience and test the platform on more patients in those real-world settings, and then go back to the US and seek FDA approval as a second stage. Or, we could take the road less travelled, and go straight to the US as our first market and negotiate FDA approval.’
It was this latter option that IDx took, and Abramoff, who is a clinical ophthalmologist, spent eight years working with FDA on how to validate the platform and how to design and test the algorithm, demonstrate that the system was safe, efficient, effective and equitable, and that it would negate the potential for racial bias.
The algorithm underpinning the IDx-DR platform is designed to mimic how clinicians might look at images. ‘They look for certain biomarkers, and so we build systems that mimic that. But, because we use deep learning very directly, we can go way deeper and pry apart what is actually going on with the disease at the finest level, to mimic how the physician analyses the images.’
Importantly, the clinical trial assessing the IDx-DR system wasn’t designed to directly compare a clinical diagnosis made by autonomous AI with a clinical diagnosis made by an ophthalmologist, Abramoff continued. Rather it was designed to measure patient outcomes. ‘After all, that’s the bottom line,’ he stressed. ‘Patients don’t want to know if the AI system is better than an ophthalmologist, they want to know if it’s going to improve their lives.’
In reality, long-term, chronic conditions such as diabetic retinopathy, progression may take years and be hard to predict, so the IDx-DR AI system was evaluated using surrogate markers of diabetic retinopathy – akin to a doctor measuring cholesterol levels as a surrogate marker for the future risk of cardiovascular disease – rather than compare it directly with a physician-made clinical decision. ‘That’s pretty unique for AI – though standard in drug trials – because physicians are almost never validated against outcomes.’
Abramoff believes truly autonomous AI could address three of the major issues with healthcare today; quality, cost, and access. ‘Autonomous AI can increase productivity and quality, drive down prices, and improve access because you can now have a diagnosis or therapy in environments where healthcare is unevenly distributed.’ Every other AI system approved by FDA to date has been an assistive AI technology, he pointed out, but while assistive AI can aid in image evaluation or assess other patient data, there is still the need for a healthcare professional or specialist to make that final clinical decision. ‘At the end of the day this doesn’t address issues of cost and access, or, indeed, quality.’
IDx then had to work with bodies including the American Medical Association, the American Academy of Ophthalmology, and many other organisations in the healthcare system to set up a payment process. There had never been a situation where a CPT code, which is required for billing, had been issued for a process without a human element, Abramoff noted. The consideration was, then how do you value the time and expertise of machines and software? ‘Fortunately, within eight weeks we had at least achieved a temporary bridging CPT code so that providers could bill and pay for use of the IDx-DR system, but it took months of discussions before we were granted a CPT code in May 2019. It was as big a hurdle to overcome as FDA blessing.’
American Diabetes Association acceptance of the IDx-DR platform diagnosis as part of standard of care for people with diabetes is also a ‘huge’ milestone Abramoff noted. ‘It often takes 8 to 10 years for new technology to become part of a standard of care. Importantly, the system has been well received both by patients and the medical community, despite initial concerns that the use of autonomous AI diagnosing could put clinicians out of jobs. ‘Which is why, 10 years ago, I was given the nickname ‘the Retinator,’ Abramoff acknowledged.
Today, the firm is witnessing mass adoption of the IDx-DR platform, and is rolling out the platform. ‘We are in an exciting phase of getting the system out there as rapidly as we can, so that it can be broadly implemented.’ In parallel, IDx-DR is working to develop additional autonomous AI systems that will bring specialty healthcare to the point of retail and primary care, Abramoff explained.
‘We want to empower primary care physicians to do more for their patients using AI, where they have limited expertise, and to feel safe doing these things. That’s why we focus on products that really lift diagnosing and therapeutic expertise from specialists, like me, to primary and point-of-care settings. Critically, this is not ‘glamour AI,’ which is technologically exciting and ‘cool’, but does not improve patient outcome, and which doesn’t have any follow through. We believe autonomous AI systems must be embedded in the healthcare system so they are part of the continuum of patient care.’
In parallel with rolling out the IDx-DR system, the company is also developing algorithms for detecting age-related macular degeneration (AMD) through retinal scans, and diagnosing glaucoma through the use of optical coherence tomography. The IDx-AMD, and IDx-G systems are projected for clinical trials during 2020 or 2021, Abramoff indicated. The firm is in addition working on AI algorithm prototypes for diagnosing ear infection and skin cancer risk.
Away from its own product development, IDx is using its experiences to help non-competing companies negotiate development and regulatory hurdles of other autonomous AI platforms, through what Abramoff calls an autonomous AI coalition. ‘There’s a lot of challenges that these companies will have to overcome, which we can help them with.’