Laboratory informatics is the specialised application of information technology aimed at optimising laboratory operations by the application of information technology to the handling of laboratory data and information. It encompasses four major multi-user systems: laboratory information management systems (LIMS), electronic laboratory notebooks (ELNs), laboratory execution systems (LES) and scientific data management systems (SDMS).
There is a very good reason why the use of a generic term such as ‘laboratory informatics’ is important: we need to get away from an application-centric approach and think of a fully integrated laboratory and its interaction with other company systems. The deployment of an ELN generally represents the final step in making a laboratory fully electronic, and hence raises the demand to connect up all laboratory systems. Being fully electronic and being fully integrated are two different things.
For most labs, being fully ‘electronic’ corresponds to an application-centric portfolio of ‘systems’ that were not necessarily designed to work together, and for which interoperability is hampered by the lack of standards and so has to be customised. A smart laboratory is an ‘integrated’ laboratory that is modular, based on standards, and is designed to facilitate connectivity, data sharing and collaboration.
Over the past few years, the informatics market has undergone two interesting developments; firstly, the previously separate LIMS and ELN sub-markets have started to overlap, causing a certain amount of confusion to the application-centric mind-set; secondly, mergers and acquisitions have reshaped the vendor line-up, specifically in the ELN field.
The origins of the LIMS market can be traced back several decades to the point where the increasing prevalence of computers in the laboratory, coupled with their increasing processing power, led enterprising scientists to develop simple, custom computerised workflow systems to operate in conjunction with data acquisition and data processing. In the early 1980s, first-generation commercial LIMS started to appear, usually based on minicomputers, supporting sample and test management, and reporting of results.
A second generation of commercial LIMS started to appear in the late 1980s, typically taking advantage of relational databases to provide more sophisticated functionality. The development of client-server based systems represented the next (third) generation of commercial systems, taking advantage of the evolution of the personal computer. The fourth generation emerged as the internet and wireless connectivity developed, offering opportunities to extend the reach of LIMS beyond the confines of the laboratory.
As LIMS products were increasingly adopted by laboratories, three specific additional requirements gradually became apparent. Firstly, there was a need to transfer data from laboratory instruments directly to the LIMS, to avoid transcription errors; secondly, the need to manage the instrument data files from which data stored in the LIMS was derived; and thirdly, the need to handle unstructured data, graphical data, and to collate sample data. These requirements led to the development of scientific data management systems (SDMS) and electronic laboratory notebooks (ELNs). Functionally, the LIMS products have become increasingly sophisticated, to the point that the dividing line between LIMS and other informatics products has become less clear.
The ELN market has grown and developed rapidly over the past decade, it still exhibits some instability with a large number of vendors (there are more than 30 purveyors of products that purport to be an ELN) competing for market share. As a consequence, the market suffers from some degree of ‘hype’ (see Figure 4).
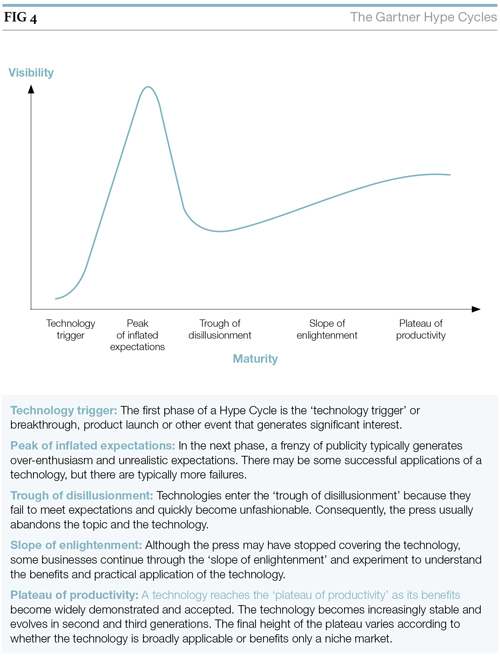
Just where ELNs sit on the Gartner Hype Cycle [1] is probably somewhere around the ‘Trough of Disillusionment’, although individual vendors may occupy positions either side of this point. The ‘Trough of Disillusionment’ can be considered as the turning point past the hype and when the focus is on delivering true benefit. Chemistry-based and generic ELNs are probably already beyond this point, as indeed are the majority of LIMS products.
Commercial ELNs have evolved from two approaches: discipline-specific; and generic. Generic software provides the architecture and tools to create and search content, and to work collaboratively in a way that satisfies the needs of almost any science-related industry. Discipline-specific ELNs are aimed at a particular market segment, such as chemistry, biology, or analytical. These systems are usually tailored to work with other discipline-specific software tools. Most of the commercial ELNs offer a combination of generic and discipline-specific functionality.
The initial evolution of the ELN market was centred on the provision of functionality to support small-molecule chemistry. Most of the experimental processes associated with synthetic chemistry are well established, reasonably consistent, and are well supported by desktop software tools. Integrating these functions into an ELN that can create, manage and store a full experimental record was a logical progression. As a consequence, chemistry-based ELNs are well established and exhibit a good deal of maturity. If there is segmentation in this part of the market, it is determined to some extent by the origins and scope of the available products. Some, for example, will be an enterprise-wide solution, others will focus on utility and personal productivity, while others will provide a generic ELN capability that integrates third-party software tools.

Biology, however, has presented a bigger challenge to the ELN vendors. The diverse and complex nature of biological processes and outcomes creates a need to capture not just the data, but also the interrelationships between the data. This, coupled with a diverse portfolio of biology-specific software tools, begs the question: do biologists just need a generic ELN that will integrate with their existing software tools, or do they need a complete suite of functionality that is embedded in the ELN? The issue for the biologists is whether there is a commercial ELN that addresses their specific and diverse requirements. Furthermore, for those companies that need to support chemists and biologists, the question is whether it is possible to find a single vendor solution that addresses the requirements of both disciplines, or whether to choose the best of breed for each discipline.
Within the past two or three years, another ELN domain has emerged, that of QA/QC and the regulatory world. A few vendors have concentrated specifically on this area, with products that are strongly aligned to laboratory workflows, following the step-by-step execution of SOPs or test methods.
The products are more structured than a ‘conventional’ ELN and in some respects appear to be functionally closer to a LIMS. This particular segment of the market has seen a number of LIMS vendors extending the functionality available in their LIMS products to embrace some of the more unstructured requirements associated with experimentation. It could be argued that such products may be better labelled as laboratory execution systems (LES) as they follow a very prescriptive approach applicable to those communities engaged in regulatory based testing.
To replace a paper notebook, all that could be required could be a simple authoring tool capable of generating a compound-document. However, additional capability will be needed for storing and searching documents, and for addressing workflow requirements. Some organisations have chosen to implement generic ELN functionality within the framework of their standard IT tools, such as Lotus Notes and SharePoint. In the academic community, blogging tools have been used to record experimental work and thus provide the basic features of an ELN, with a strong emphasis on sharing and collaboration and in the form of a laboratory journal.
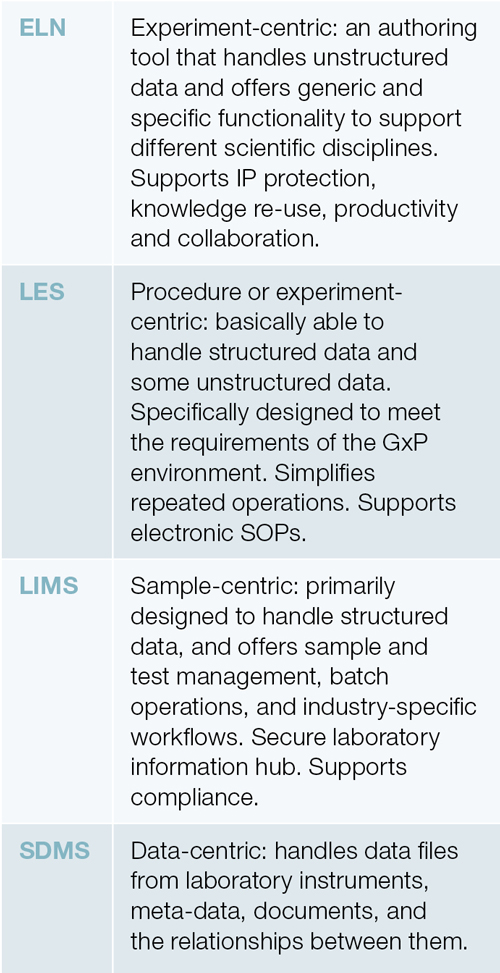
The convergence in the informatics market is now confusing potential customers. The table, right, identifies the core differences in the major tools.
Initially, each of these tools addressed a well-defined, functional requirement, but the increasing level of sophistication of the underlying information technologies has made it easier to extend functionality in ways that mean that there is now considerable overlap between the different tools. At one stage it was considered unlikely that a single ELN could provide the necessary functionality to support chemistry, biology and analytical requirements. Those days are over, and this should make the task of finding a suitable ELN easier. But the extent of the overlap with LIMS, SDMS, and LES can generate confusion, and for someone looking to address laboratory information management requirements, the task seems to be more challenging.
What is a laboratory information management system (LIMS)?
A laboratory information management system (LIMS) provides the basic functions for sample and test management, and has become the standard tool for analytical and QC laboratories for registering samples, assigning tests, gathering and managing results, and issuing reports. Most LIMS now provide a more integrated solution to support workflows and processes customised to a range of industry-specific requirements.
The basic functions to be found in a LIMS are:
- The registration of samples and associated data, such as provenance, customer, due dates, etc.;
- The assignment of tests to the sample;
- Scheduling and tracking of the sample and tests;
- Recording the test procedure, equipment and materials used during testing;
- The review, approval, and aggregation of test results for the sample, including specification checking ; and
- The preparation and communication of customer reports.
The major business benefits of a LIMS are typically associated with more efficient workflows by eliminating errors due to manual data entry and transcription errors. This is achieved through interfacing laboratory instruments to the LIMS for two-way communication of sample IDs, worklists, and results, and by integration with other laboratory systems such as electronic laboratory notebooks (ELNs) and scientific data management systems (SDMS).
A LIMS also acts as a major repository of the records of analytical testing and can be a source of historical data associated with the organisation’s products and production processes. In addition, the transactional nature of a LIMS enables a secondary record system to be maintained as an audit trail to track date, time, user – and, if necessary, what change was made within the system. This data may then be used to satisfy quality assurance requirements in terms of data integrity, and can also be used to generate a wide variety of management reports on the laboratory’s performance.
A pre-requisite before implementing a LIMS, or indeed any major computerised system, is to map and optimise the laboratory processes that the LIMS will automate. The laboratory needs to understand the process and to identify any bottlenecks and their underlying causes. Most laboratory processes have evolved over time to meet local laboratory requirements rather than being specifically designed to meet wider organisational requirements. Any LIMS implementation must simplify and streamline the process rather than automate an inefficient, paper-based status quo.

The commercial systems on the marketplace have become increasingly sophisticated over the years. The major challenge in choosing a LIMS is identifying how an out-of-the-box solution is aligned to the organisation’s needs. Most systems are highly configurable and avoid the need for any custom code to be written to meet specific requirements.
What is a scientific data management system (SDMS)?
A scientific data management system (SDMS) is, in its basic form, a database application that manages electronic records generated by laboratory instruments. Typically, an SDMS will provide long-term data preservation, accessibility and retrieval. It is complementary to other laboratory informatics systems, such as LIMS and ELNs, in the sense that it can provide a common repository for experiment- and sample-related data files. In this way it provides a more consistent approach to managing laboratory data than local repositories, and off-line media (CDs, DVDs, tape, etc.)
The lines between a LIMS, ELN and an SDMS are at times blurred through the incorporation of additional features to complement the core functionality. An SDMS is a means of collecting data files from a wide range of laboratory instruments and storing them, along with metadata, in a uniform way in a database; in other words, it is a laboratory content management system. By adding workflow elements and providing facilities for the management and storage of other documents associated with laboratory operations (worksheets, SOPs, safety information, reports, PDFs, office documents, images, etc.), an SDMS can in practice evolve into a more comprehensive single informatics solution for some laboratories. However, an SDMS is essentially an ‘event-driven’ system that gathers data, which may limit some of its capabilities relative to the other informatics tools, and is therefore more frequently seen as a system that is complementary to a LIMS or an ELN.
Nevertheless, the principle on which the SDMS is based is that it aggregates records into a logical collection associated with a specific entity such as a programme, project, experiment, product, or sample, to provide a readily accessible collection of relevant information. Embedded into an SDMS will also be the means to provide appropriate security of the records by means of access control, audit trail, authorisation, and change management.
What is a laboratory execution system (LES)?
A laboratory execution system (LES) sits somewhere between an ELN and a LIMS in terms of the functionality it delivers, but its existence is typically targeted at analytical service and quality control laboratories where high-volume workflows and regulatory compliance are primary business requirements. In a very basic sense, the underlying logical structure of an LES is almost identical to a LIMS, but the user interface is procedure-centric, rather than the usual sample-centric approach found in a LIMS. This allows a standard laboratory operating procedure (SOP) to be executed in an automated way, usually by interaction with laboratory instruments interfaced to the LES (where possible) in order to capture data without the need for transcription. Calculations on the captured data can be performed in the system, and thus the automated approach can eliminate two potential sources of error. The concept of a ‘paperless lab’ is a specific objective of the LES, eliminating the use of paper either for intermediate recording of data, or for longer term record keeping and archival purposes.
The LES is designed to adhere to laboratory workflows and provides a more repeatable and structured approach to quantitative testing procedures to help ensure compliance. The user interface usually takes the form of an electronic equivalent to the paper version of a laboratory standard operating procedure or worksheet. This type of interface is often referred as ‘paper on glass’, a term also used for a generic electronic laboratory notebook. Most LES applications can be readily configured to support alternative laboratory workflows in a way that relates closely to traditional paper-based processes. Worksheets can be converted to electronic forms; standard operating procedures can be configured with appropriate data entry points, with data checking; and unique workflows can be mapped to support repetitive and routine procedures.
As with other laboratory informatics systems, the underlying information technologies can extend an LES to a broader range of capabilities. For this reason, the LES can, in some cases, serve as an alternative to a LIMS, an ELN, or an SDMS. As with each of the major laboratory informatics tools, purchasing and implementation decisions require a thorough understanding of the laboratory’s functional requirements. However, it is more likely that the LES will be seen as complementary to ERP and QM systems where high-throughput QA is an essential step in a business process.
What is an electronic laboratory notebook (ELN)?
In its simplest form, an electronic laboratory notebook can be considered to be a direct replacement for the paper lab notebook. In this instance, it can provide the generic functionality (‘paper on glass’) to support scientific documentation for patent evidence, cross-discipline collaboration, and general record keeping. However, the integration capabilities raise the possibility of a tighter coupling of other laboratory systems into the ‘electronic laboratory notebook’. In other words, can the information that is currently printed from other laboratory systems, cut out and pasted into the paper lab notebook, be electronically entered or linked directly to the electronic laboratory notebook?
For example, systems that provide chemical structure drawing, structure and sub-structure searching, and compound registration are an integral part of the chemistry laboratory’s process, and therefore would be expected to become part of an electronic solution. Similarly, other scientific disciplines will have specific requirements consistent with their particular laboratory processes.
Figure 5 illustrates the relationship between ‘broad’ (generic) and ‘deep’ (specific) systems. In this context, the ‘notebook’ functionality (see Figure 1) is addressed by the ‘broad’ layer, whereas the discipline-specific functionality penetrates the ‘interpreted/processed data’ layer in Figure 1.
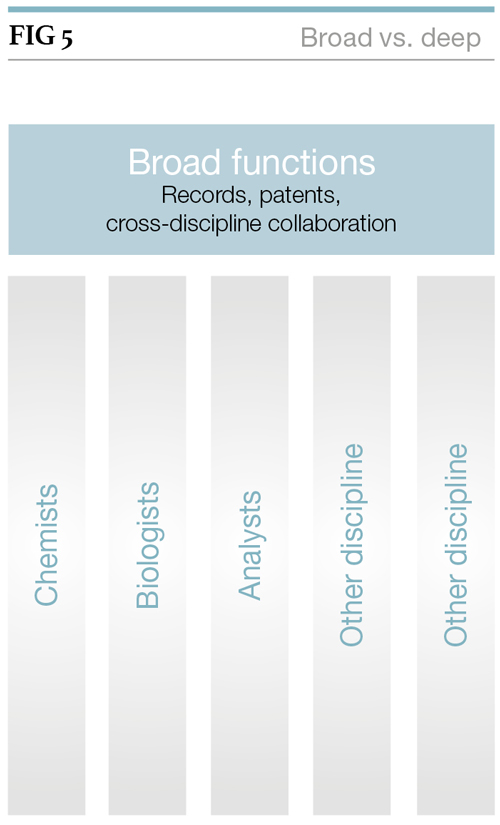
From a patent perspective, the ‘experimental layer’ of Figure 1 is crucial as it captures what the scientist is thinking and doing, and therefore will provide the evidence of conception and reduction to practice of the ‘invention’. In broader intellectual property (IP) terms, it is the ‘experiment’ layer that constitutes a record of the laboratory’s work and as such contributes to the scientific knowledge repository.
For as long as this repository resides on paper, the ability to access, collaborate and share scientific knowledge is constrained. The implementation of an ELN therefore offers a significant opportunity to bring about greater efficiencies.
But a clearly defined understanding of the role that the ELN is going to play in a given organisation is absolutely essential at the start of an electronic laboratory notebook project. As discussed, an electronic laboratory notebook supports the ‘experiments’ layer, and also contains abstractions from the lower data levels (see Figure 1).
So the CENSA [2] definition of an electronic laboratory notebook as ‘a system to create, store, retrieve and share fully electronic records in ways that meet all legal, regulatory, technical and scientific requirements’ is all encompassing and can mean different things to different people.

An ELN can serve the organisation in three ways: it can take advantage of the capabilities of IT to improve the ability to acquire, manipulate, share and store data (productivity); it can facilitate communication and sharing in real-time across multi-disciplinary and multi-site teams (collaboration); it can provide a scientific knowledge repository that can be easily accessed to recover records of the laboratory’s work (content/knowledge management).
The way in which laboratory notebooks are used is largely dictated by the United States’ patent system which, unlike the rest of the world, is based on ‘first to invent’. The need to be able to demonstrate who really was first to invent requires the laboratory notebook to be an authentic and trustworthy record that describes the concept and its reduction to practice, and for it to be signed by the author and corroborated by an impartial witness. There are two factors why the migration away from paper lab notebooks has taken so long: the reluctance of lawyers and patent attorneys to gamble on the legal acceptance of electronic records in patent interferences and patent litigation without any case law; and the lack of confidence in our ability to preserve electronic records over several decades.

One of the challenges to a successful ELN implementation is identifying exactly what role the ELN will play. The term ‘electronic laboratory notebook’ is inherently ambiguous. In most cases, the ELN is expected to do more than just replace the paper lab notebook. The paper lab notebook is a simple authoring tool, and any electronic authoring tool capable of generating a compound document will serve as a replacement.
For some companies this has proven to be the case. The combination of Microsoft Office, SharePoint services and a means of preserving documents (e.g. in PDF – portable document format) has proven to be an adequate replacement for paper. But if more functionality than this is needed – for example, integrating various chemistry or biology-centric functions, or other discipline-specific tools – then we are really talking about an electronic laboratory rather than an electronic laboratory notebook.

Chapter summary
The four major laboratory informatics systems serve different basic functional requirements, but convergence and increasingly sophisticated technologies are creating a good deal of overlap between the systems.
So when it comes to choosing the right solution, it’s far better to start by defining an objective or describing the problem to be solved, rather than placing the initial focus on a ‘solution’.
Just deciding ‘we need an ELN’ or ‘we need a LIMS’ should not be the starting point; it’s far better to think about the big picture, i.e. the end-to-end business process that embraces the role of the laboratory, the specific workflows, the communication and collaboration requirements, and the integration requirements.
Once these requirements are defined, then the task of finding a solution is more straightforward.
Next: Knowledge: Document management >


